Selected Case Studies
01
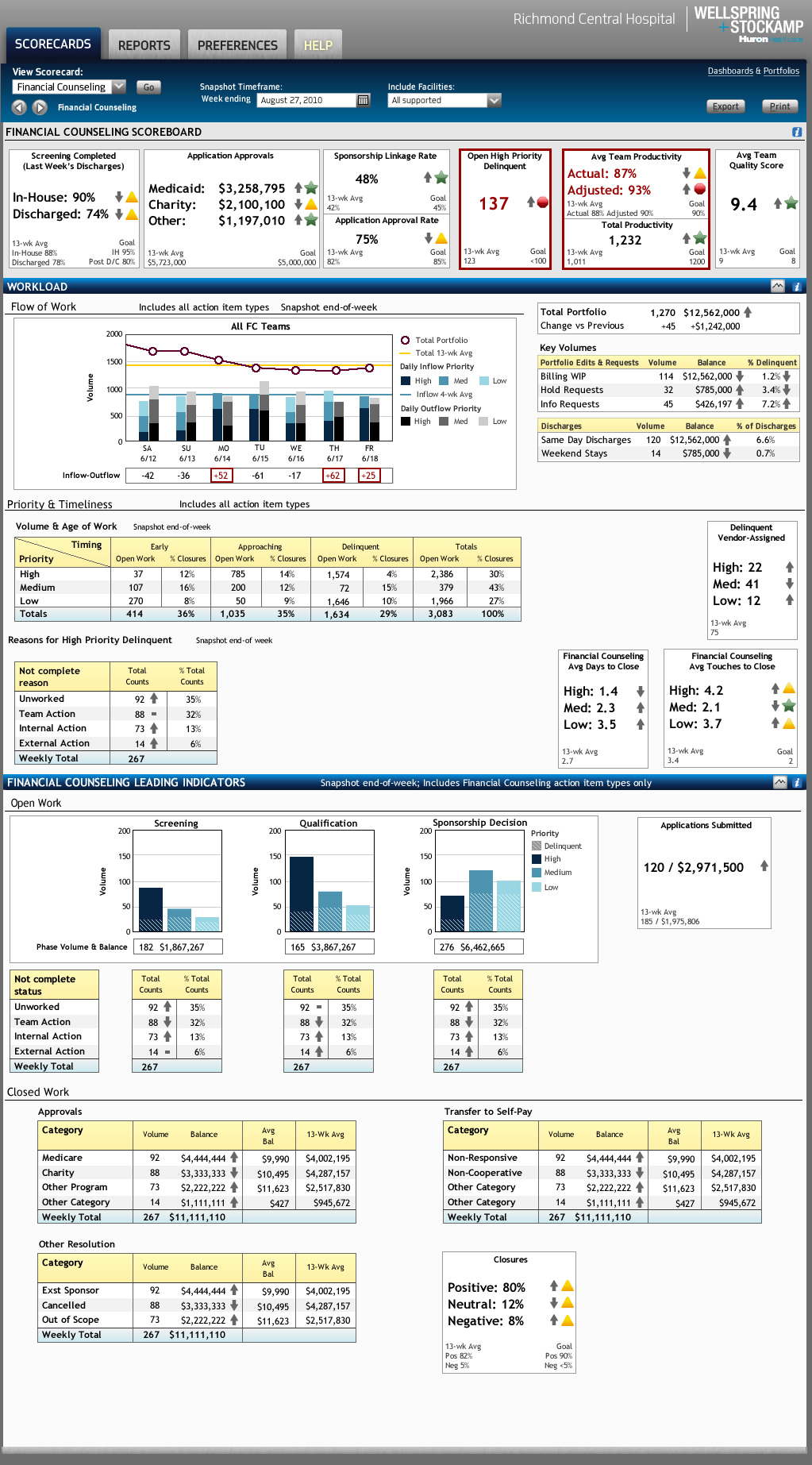
Huron Consulting
Hospital REVENUE CYCLE MANAGEMENT SOFTWARE
How field research across a complex, multi-role hospital workflow produced a unifying design insight — and an enterprise platform large enough to require three simultaneous consulting teams working in parallel.
Services: User Research ▪︎ Interaction Design ▪︎ Information Design ▪︎ Visual Design ▪︎ Business Intelligence Design ▪︎ Design Leadership / Product Management
02
Ellipsis Health
AI/ML-powered Mental Health APP
How a ground-up UX redesign transformed Ellipsis Health’s voice-based mental health platform from a confusing experience into a product that impresses clinicians, converts investors, and guides patients with care.
Services: UX Redesign ▪︎ Interaction Design ▪︎ Prototyping ▪︎ Usability Testing ▪︎ Design Sprint ▪︎ Product Management
03
Fluke Networks
INDUSTRIAL CABLE ANALYZER TOOL
How deep field research and a project-centric design approach turned a hardware refresh into a market-leading product.
Services: User Research ▪︎ Interaction Design ▪︎ Visual Design ▪︎ Prototyping ▪︎ Usability Testing
04
St. Jude Medical
Class III Medical device platform
How recentering a Class III medical device around its true primary user — the Pacer Nurse — transformed St. Jude Medical Cardiac Rhythm Management Division’s market position and produced a product the industry lauded for years.
Services: User Research ▪︎ Interaction Design ▪︎ Usability Testing ▪︎ Human Factors Engineering ▪︎ Product Management

Testimonials
Read what some satisfied clients shared about working with Devise…